
For Al through to Ar, first ionisation involves removal of an electron from the 3p subshell. In Na and Mg, first ionisation involves removal of an electron from the 3s subshell. As a result it requires an increasing amount of energy to remove an outer shell electron across the group. There is a generally increasing trend in ΔHi 1 across Period 3, although we also see a small dip after Mg and again after P, so we need to be able to explain why ionisation of Al and S requires a little less energy than might be expected.Īcross a period: Across a period the electron being removed in first ionisation comes from the same shell, and the outer shell electrons experience the same shielding from filled inner shells, but the number of protons increases and hence the nuclear attraction towards the electron being removed increases. There are clear patterns in the ionisation energies across one period compared to other periods – compare Period 2 (red) and Period 3 (green), for example, and note the same pattern in Period 4 (purple) if the d-block elements (grey) are ignored. ii) Properties that depend on electron arrangement: first ionisation energiesĬonsider the first ionisation energies of the first 40 elements: Period 4 shows the same periodic trend too, although the pattern is there interrupted by the filling of the 3d subshell after 4s is filled. Exactly the same pattern is seen across Period 2 with the filling of the 2s and 2p subshells 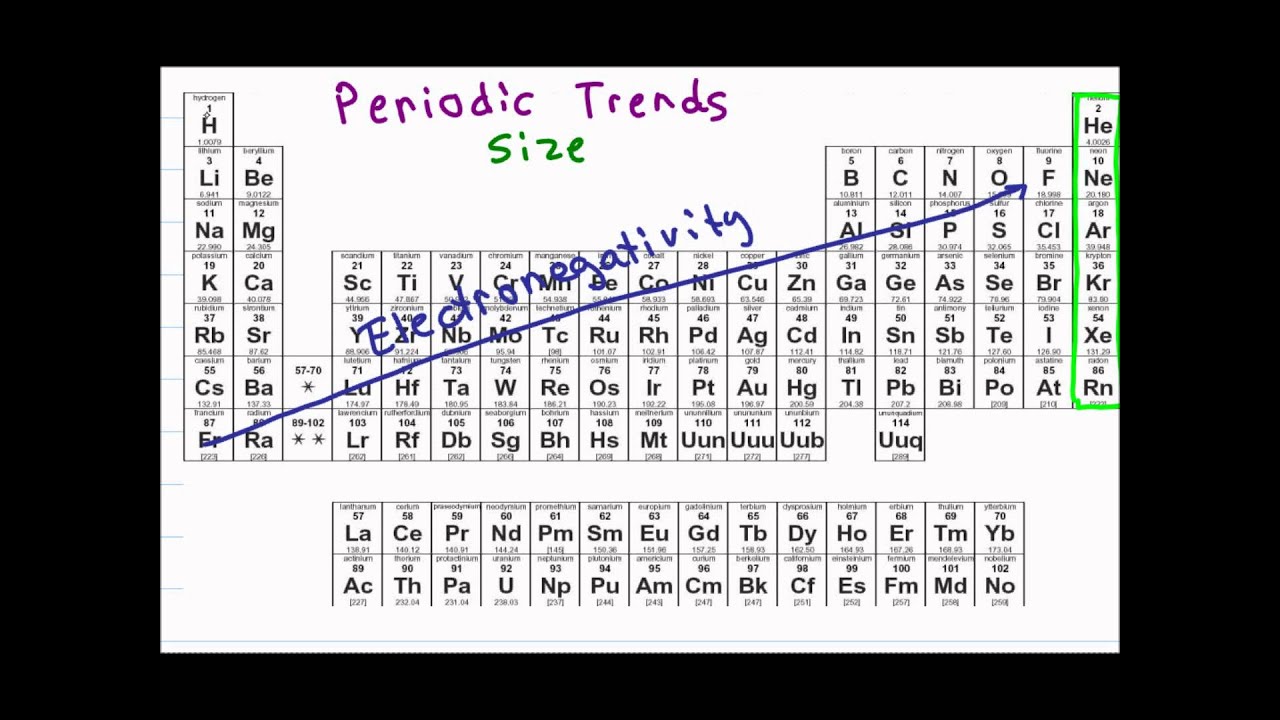
Electron arrangement i) Electron arrangements in Period 3 NaĪcross Period 3 we see an increasing number of electrons in the n=3 shell, with the 3s subshell being filled first, followed by the 3p subshell. Using Period 3 as an example, we can start to examine some periodic properties that show the same patterns and trends across other groups. This also means that there are repeating trends and patterns in physical and chemical properties from one period to the next, which we refer to as periodicity. The consequence of this is that elements in each Group (column) have the same number of outer shell electrons and therefore similar physical and chemical properties. The modern periodic table is arranged in order of increasing atomic number (proton number), with a new Period (row) started every time electrons occupy a new shell. 
These he attributed to elements that had not yet been discovered, and he went on use his table to successfully predict the properties of these undiscovered elements based on the properties of known elements in the same Group. Mendeleev’s organization of the periodic table into atomic weight order, with elements having matching properties being placed in the same column, required him to leave gaps where the properties did not match. Newlands was one of the first chemists to describe a repeating pattern in physical and chemical properties when the elements were placed in order of atomic weight – his Law of Octaves, although it did not work for all elements, was the first periodic law.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
